
For example, the standard molar entropy of some solids are given below: (white) (rhombic)ĭiamond graphite sodium phosphorus sulfur silverĢ.38 5.74 51.3 41.1 31.8 42.6 J (K mol) -1 They have been carefully measured for many substances. 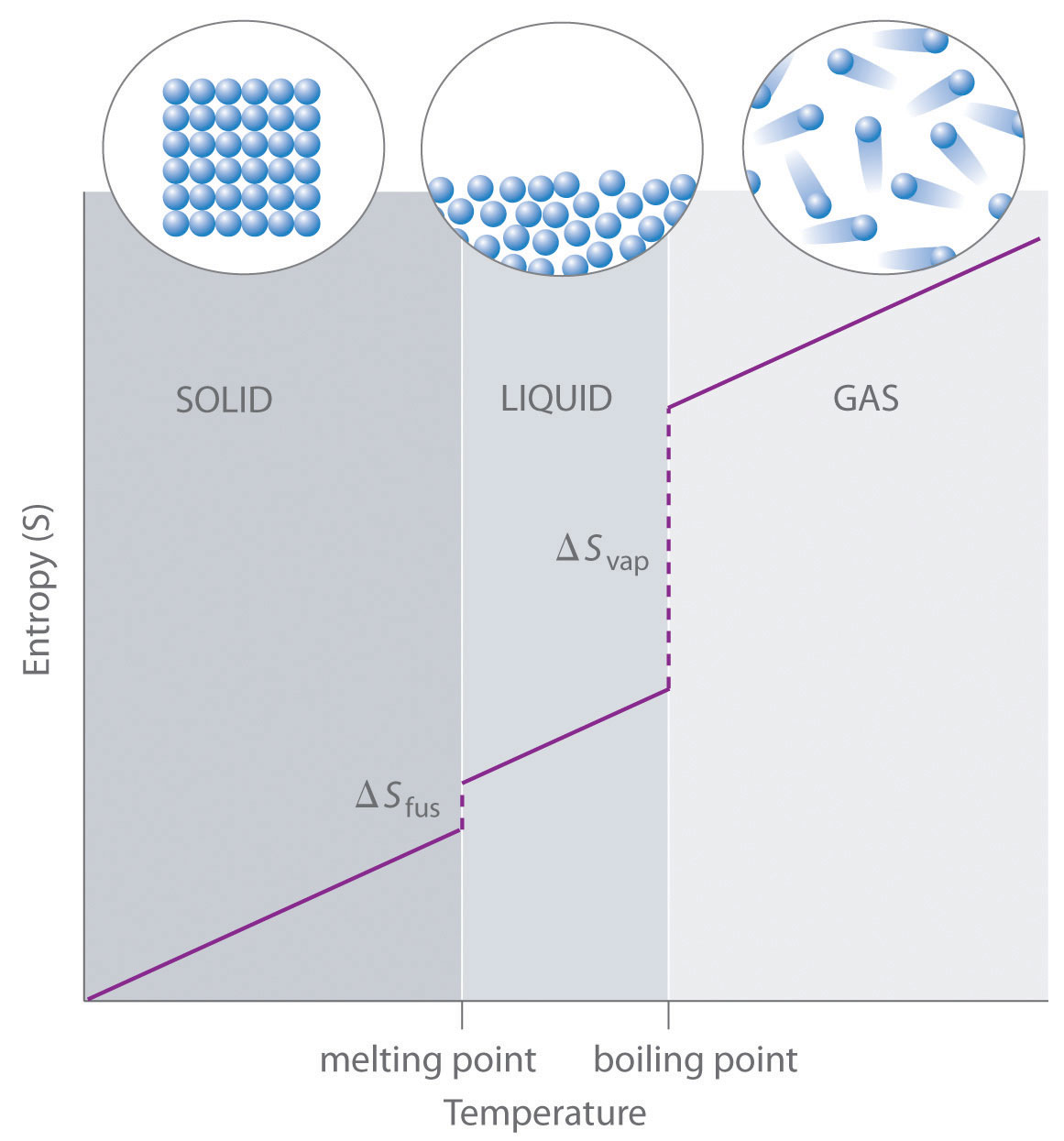
Thus, the entropies S so evaluated T = 298 K are called standard entropies. Recall that thermodynamic values at standard condition are called standard values. In a phase transition, heat is absorbed, but the temperature remain constant. Of course, the heat capacity C may also vary with temperature. The area below the curve of a plot of q / T versus T from 0 K to T is then the absolute entropy S of the system. A substance is in a completely ordered crystalline state, at which the moleules contains no removalbe vibrational, rotational, translational, or even thermal disorder energy.Īs energy q is absorbed by a substance, its temperature increases by dT. At absolute zero Kelvin, the substance contains no removable energy. Entropy contained in a system, say in a mole of a pure substance, is a theoretical quantity that takes account of all heat transferred to it since the lowest atainable temperature, 0 K. Sorry for being so formal, but just so that you know you know something classical.īy definition, the change in entropy can be evaluated by measuring the amount of energy transferred. This statement is one of the acceptable statement of second law of thermodynamics. Thus, the driving force for a spontaneous process in an isolated system is an increase in entropy. Such a change is called a spontaneous process. Nature has a tendency for entropy S to increase, and the system changes in response to this tendency. If the change takes place quickly in an irreversible manner, the entropy is greater than what is evaluated, because the temperature increase is not uniform. 
However, the changes are supposedly take place slowly over a long period of time, or in an almost equilibrium or reversible condition. You have learned the concept of integration in a calculus course.Įntropy is a state function in that it depends only on the initial and final state of the system, regardless of the path by which the changes take place. This sum can take the form of integration if the temperature various contineously. If the process takes place over a range of temperature, the quantity can be evaluated by adding bits of entropies at various temperatures. Thus, entropy has the units of energy unit per Kelvin, J K -1. When a system receives an amount of energy q at a constant temperature, T, the entropy increase D S is defined by the following equation.Įntropy is the amount of energy transferred divided by the temperature at which the process takes place. Entropy is related to the energy distribution of energy states of a collection of molecules, and this aspect is usually discussed in statistical mechanics. Traditionally, the entropy concept is associated with the second and third laws of thermodynamics. These changes cause an increase in entropy for the system under consideration, but energy is not transferred into or out of the system. A different concept is required to explain spontaneous changes such as the expansion of a gas into an abailable empty space (vacumm) and heat transfer from a hot body into a cold body. Human experienced chemical and physical changes that cannot be explained by energy alone. This concept was developed over a long period of time. Entropy, symbol S, is related to energy, but it a different aspect of energy. We have define energy as the driving force for changes, entropy is also a driving force for physical and chemical changes (reactions). We confine our discussion to thermodynamics (science dealing with heat and changes) and to chemical and physical processes. The word entropy is used in many other places and for many other aspects. You are not alone if you have some difficulty with this concept. Thus, few people understand what entropy really is. In all other cases the system (the gas) is restored to its initial state, but the surroundings are forever changed.Ī reversible change is one carried out in such as way that, when undone, both the system and surroundings (that is, the world) remain unchanged.\)Įntropy is a chemical concept that is very difficult to explain, because a one-sentence definition will not lead to a comprehensive statement. In the limit of an ”infinite” number of steps (bottom), these work terms are identical, and both the system and surroundings (the “world”) are unchanged by the expansion-compression cycle. As the number of steps increases, the processes become less irreversible that is, the difference between the work done in expansion and that required to re-compress the gas diminishes. These diagrams show the same expansion and compression ±ΔV carried out in different numbers of steps ranging from a single step at the top to an "infinite" number of steps at the bottom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
